Dale Gort
ENGINEERING & PRODUCT LEADER - TAKING COMPLEX PRODUCTS FROM CONCEPT TO FULL-SCALE PRODUCTION
ENGINEERING & PRODUCT LEADER - TAKING COMPLEX
PRODUCTS FROM CONCEPT TO FULL-SCALE PRODUCTION

When a Product Moves to Production — Where Success Is Critical
You have an early product concept that works.
Engineering is confident.
The opportunity is clear.
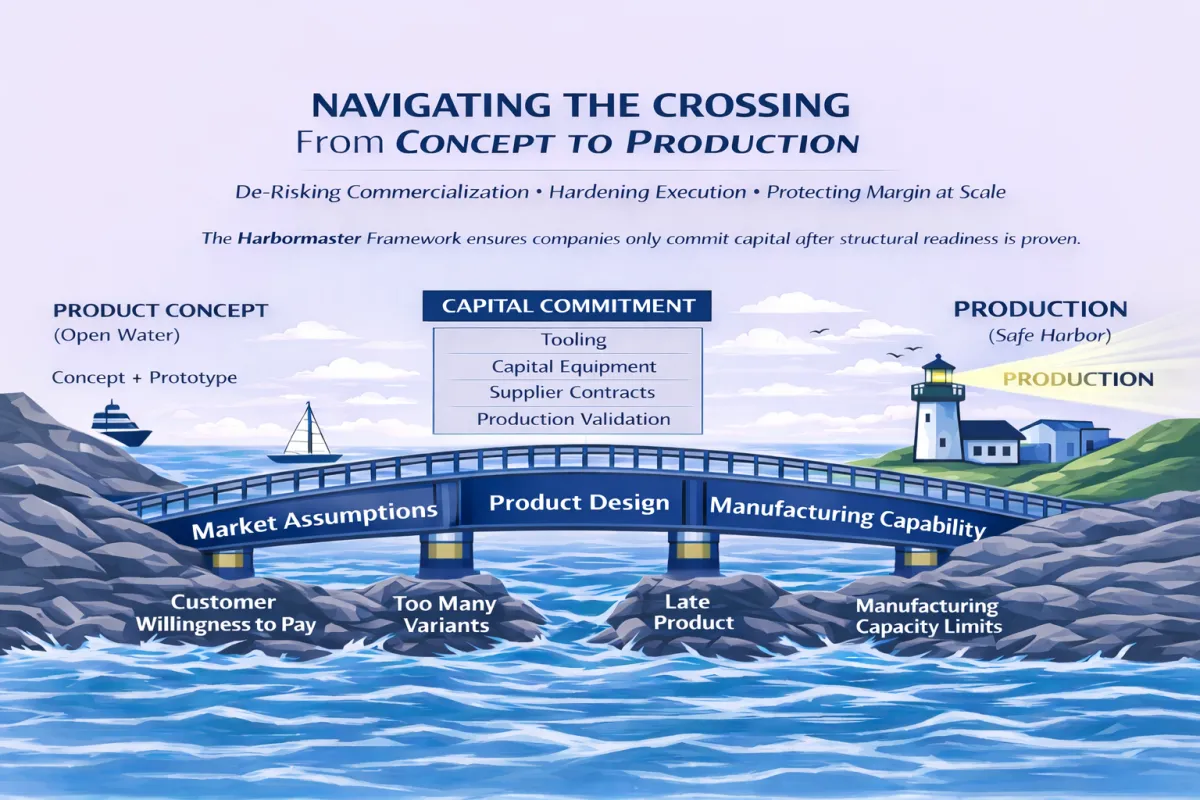
But the bridge from early product concepts to production introduces a different kind of complexity.
This is the point where disciplined execution matters most.
THE SITUATION
I get called in at different stages, usually when things start to stall.
The most advantageous time is at the beginning, before misalignment sets in and small issues become expensive.
Most new product launches don’t fail in just one area. The fail because of gaps as they navigate the bridge to production:
Launch Teams are not aligned
Scope creep in the product definition goes unchecked
Plans exist, but don’t hold up under pressure.
Timelines slip without a clear cause.
Capital is going out, but progress is unclear.
The issue isn’t effort or lack of talent. It’s lack of structure in how the bridge to full production is crossed.
THE APPROACH
I step in to help companies navigate the crossing from early-stage concepts to full production with clarity and control.

Executive Alignment
Keep leadership aligned as commercialization decisions accelerate.

Cross-Functional Readiness
Ensure design, manufacturing, and marketing stay synchronized.

Assumption Validation
Test the assumptions behind cost, scale, and supplier capability.

Disciplined Progression
Ensure scale decisions follow verified readiness — not timeline pressure.
Early-stage Products to Production Alignment
I align engineering, manufacturing, suppliers, and leadership around a plan that can be executed.
Commercialization & Launch Execution
I step into critical programs that are drifting and bring them back under control.
Risk & Readiness Assessment
The work centers on one objective:
Establish a controlled, reliable bridge from early-stage concepts to full-scale production.
Where Harbormaster™ System Operates
This is where the Harbormaster system is applied—in the crossing, where decisions start to carry real consequence.
WHAT THIS ENABLES
With the right structure in place:
Teams align around a single execution path
Risks are identified and addressed early
Progress becomes measurable and predictable
Leadership gains clear visibility and confidence
The result is not a just a successful launch, but a repeatable path.
WHAT THIS PROTECTS
When commercialization breaks down, the cost is not just time.
Missed revenue targets
Capital tied up in the wrong decisions
Delayed market entry
Loss of confidence at the leadership or board level
My role is to prevent those outcomes by bringing clarity, alignment, and execution when it matters most.
If you are approaching production—or already in it— and need a clear, controlled path forward, this is the point to engage.
Selected Results
Greenfield Facility Launch
Led a manufacturing facility from construction to full production in 5 months, building a 150-person operation and achieving run-at-rate performance.

$80M Manufacturing Investment Assessment
Evaluated facility design, production flow, and capital requirements to ensure scalability before investment.

NIH/FDA Program Scale-Up
Advised diagnostic companies scaling to 50 million units per month under federal oversight, resolving production and regulatory bottlenecks.
$20M+ Product Launches
Led cross-functional teams to bring complex products to market with aligned engineering, manufacturing, and supplier execution.

FOR PRIVATE EQUITY & ACQUISITIONS
I support investment teams and operating partners at two critical points.
Pre-Acquisition
Assess whether a product can be manufactured, scaled, and delivered profitably before capital is committed.
Post-Acquisition
Stabilize product, manufacturing, and execution issues so portfolio companies can hit production targets and protect value.
Focus areas:
Product scalability and manufacturability
Execution risk and timeline credibility
Validate assumptions before they become sunk cost.
Capital efficiency and operational readiness
TYPICAL SITUATIONS I’M BROUGHT INTO:
Stabilizing a Post-Acquisition Product Redesign
Brought in after acquisition to align product, regulatory requirements, and manufacturing readiness, enabling entry into new markets without delay.
Scaling a High-Risk
Federal Program
Worked with NIH and FDA-backed companies to remove production bottlenecks and accelerate time to market under intense public and regulatory pressure.
Recovering a Delayed
Product Launch
Realigned engineering, suppliers, and production planning to stabilize timelines and protect revenue commitments.

About Me
Great products do not fail because of bad ideas.
They fail in the transition from design to production — where complexity increases, alignment breaks down, and small issues become expensive problems.
That is where I work.
I have led complex product and manufacturing programs across automotive, medical diagnostics, advanced manufacturing, and building products — environments where precision matters and the margin for error is thin.
Selected experience:
Led launch of automotive greenfield facility from construction to full production in 5 months
Directed $80M manufacturing investment assessment
Delivered product launches tied to $20M+ revenue targets
Advised NIH/FDA-backed programs scaling to millions of diagnostic devices per month
I am brought in when the path to production is unclear — and the cost of getting it wrong is high.
If the Product Works — But the Business Isn’t Scaling
That is usually the moment companies reach out.

Quick Links
Legal
Copyright 2026. Shorefront Strategies, LLC. All Rights Reserved.
